China’s medical device sampling inspection plan for 2024 was issued by the National Medical Products Administration (NMPA) on March 19, 2024. This document outlines the inspection plan for medical devices during 2024 and sets forth specific requirements for its implementation. We’ve listed the key points and implications for regulatory affairs departments below:
1. Inspection Requirements:
Local Medical Products Administration (MPA) as well as other relevant institutions are mandated to organize and instruct inspection agencies to conduct inspections in accordance with the prescribed inspection plan. These inspections are to be carried out with reference to the mandatory standards and the Technical Product Requirements (PTR) of the registered or filed medical device.
All being well the inspection is successful. However, it may be the case that the inspection couldn’t be completed due to missing data or an inadequate PTR provided by the registrant or the NMPA Legal Agent of the imported product. In such instances, inspection agencies are expected to collect information on cases and the local MPA will issue a medical device sampling inspection deficiency advisory letter to the registrant or legal agent.
Failure to cooperate with the sampling inspection may result in consequences such as entries in the company’s Enterprise Credit File and intensified supervision and inspection in the future as well as a re-inspection. The results of the relevant investigation and handling will be entered into the National Medical Device Sampling Inspection Information System within 30 working days after the issuance of the advisory letter.
2. Re-inspection Requirements:
Where the inspection has not been successful, a follow-up inspection may be requested by the registrant or legal agent, this is known as re-inspection. The re-inspection acceptance department is the provincial MPA where the registrant, filer, or legal agent is located. The re-inspection agency is determined by the provincial MPA.
- Re-inspection applications for the same inspection report are only processed once
- Re-inspection applications submitted after the deadline (7 working days from the date of receipt of the inspection result) or repeat requests will not be accepted
- Re-inspection is not permitted for inspection items that are explicitly designated as risk monitoring sampling.
- After the local MPA accepts a re-inspection application and makes a re-inspection decision, the applicant cannot withdraw the re-inspection application.
3. The China Medical Device Sampling Inspection Plan 2024
The 2024 medical device sampling inspection plan includes 66 product varieties such as high-frequency electric scalpels, breast implants, Helicobacter pylori antibody test kits (boxes), medical protective masks, arthroscopes and much more.
The focus of the 2024 plan ’is primarily on medical electrical equipment, including carbon dioxide laser therapy machines, electrocardiographs, and medical pulse oximeters.
It also involves soft contact lenses, hydrocolloid dressings, titanium and titanium alloy dental implants and scar repair gels. The plan clearly specifies:
- basis of the inspection
- inspection items
- judgment principles for the product in question s
- initial inspection and re-inspection agencies.
Examples of China’s medical device sampling inspection plan for 2024:
- Inspection basis: Product Technical Requirements,
- Inspection item:
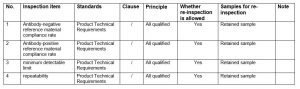 The principle of comprehensive judgement:
The principle of comprehensive judgement:
- Any one of the table is judged to be unqualified, the comprehensive conclusion of the sampling is unqualified.
- Samples in the normal inspection process can not be used normally, the sampling and testing of the overall conclusion is unqualified.
- Applicable test items in the technical requirements of the product as the basis for determination. The name of the project in this programme to issue an inspection report.
- Listed test items involved in the recommended requirements are not included in this test programme.
4. China Medical Device Sampling Inspection 2024 Results:
Registrants, legal agents, and sampled units must take immediate risk control measures upon receiving reports indicating non-compliance. Regulatory authorities are responsible for promptly investigating cases that do not meet the criteria . Any criminal cases will be transferred to judicial authorities in accordance with the law.
Further information
Read the original NMPA announcement on China medical device sampling inspection 2024.
Read our recent article on the NMPA recommencing overseas inspections here.
Contact Cisema for assistance in preparing for your NMPA overseas inspection.
GET IN TOUCH

 Deutsch
Deutsch  Italiano
Italiano  Français
Français  日本語
日本語  한국어
한국어 



