The China Drug Evaluation Report 2023 has been issued by the Center for Drug Evaluation (CDE). It provides valuable insights into the registration trends shaping the pharmaceutical industry in China. Below we present the key findings.
Unprecedented Growth in Drug Registrations:
In 2023 there was a significant increase in drug registration applications and approval rates. The CDE accepted 16898 registration applications in 2023 which is an increase of 36.6% over the same period last year, indicating a robust pipeline of pharmaceutical innovations.

Applications requiring technical review by category
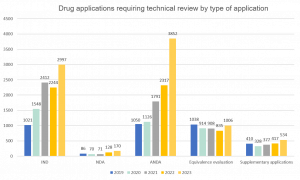
Applications for Investigational New Drugs (IND) and Abbreviated New Drug Applications (ANDA) increased dramatically to reach their highest points over the last five years at 33% and 66% growth year-on-year (YoY) respectively. Increases were also witnessed for New Drug Applications (NDA), Supplementary Applications and Equivalence Evaluations.
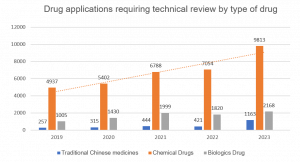
Chemical drugs remain the dominant drug modality in China, experiencing continued growth in applications at +39% YoY, and nearly doubling since 2019.
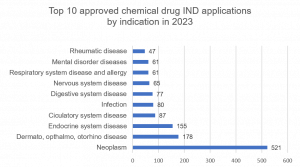
Chemical drug IND applications approved by CDE in 2023
Approved applications for chemical INDs increased by 12.6% from 1286 approvals in 2022 to 1448 approved in 2023. Approvals for neoplasm remained number one even though approvals decreased by 8%. The combined fields of dermatology, ophthalmology and otorhinolaryngology remained in 2nd place experiencing 34% growth. Interestingly, approvals for the endocrine system jumped dramatically by 260% from just 43 approvals in 2022 to 155 in 2023, reaching 3rd in the top 10. Entering the top 10 fields for approvals in 2023 were Rheumatic disease and Mental disorder diseases, while Renal/urine diseases and Autoimmune/immunosuppressant disease categories fell out of the top 10.
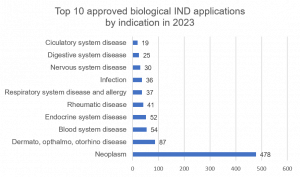
Biologic drug IND applications approved by CDE in 2022
China’s Center for Drug Evaluation approved 59 IND applications for preventive biological products, 31 of which were new innovative preventive biological products, representing a year-on-year increase of 72.2%, reversing 2022’s decline of 25%. In addition, the CDE approved 891 IND applications for therapeutic biological products, 695 of which were for innovative therapeutic biological products, which represents a year-on-year increase of 25.7%, showing increasing growth. Of the approvals that were granted, over half were for the field of Neoplasm which grew by 10% unlike those for chemical IND approvals. The combined fields of Dermatology, Ophthalmology and Otorhino disease witnessed significant growth of 81% and retained their second place in the top 10. The Endocrine system indication which witnessed dramatic growth in chemical IND approvals, also experienced growth in biologicals of 18% YoY. As for chemical IND approvals, Rheumatic disease indication entered the top 10 along with Circulatory disease biologicals, whilst Prophylactic vaccines and Autoimmune/Immunosuppressant indications left the top 10. Other indications remaining in the top 10 but experiencing significant decreases between 27% and 57% were Digestive, Infection, Respiratory and Nervous system diseases.
Expected Pathways for Drug Approval:
In 2023, the CDE shortened the time for drug development and technical review by accelerating the listing and registration process for medicines and strengthening communication and interaction with applicants. China has implemented expedited pathways for drug approval, particularly for therapies addressing critical public health needs. The report highlights the role of these “fast-track” mechanisms in accelerating approvals and ensuring timely access to life-saving medications for patients.
Alignment with International Standards:
China continues to align its regulatory standards with international best practices to enhance global collaboration and facilitate market access for pharmaceutical products. In terms of the transformation and implementation of the International Council for the Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) guideline documents, the NMPA has completed the implementation of all 68 current ICH guidelines.
Further information
Read the original report in full: China drug evaluation report 2023.
Read our post on the 2022 report about China drug registrations.
If you would like advice on the best regulatory pathway to market for your pharmaceutical, please contact Cisema.

 Deutsch
Deutsch  Italiano
Italiano  Français
Français  日本語
日本語  한국어
한국어 



